|
→ Write the subshell electronic configuration of the following elements and find the blocks to which they belong.ī. 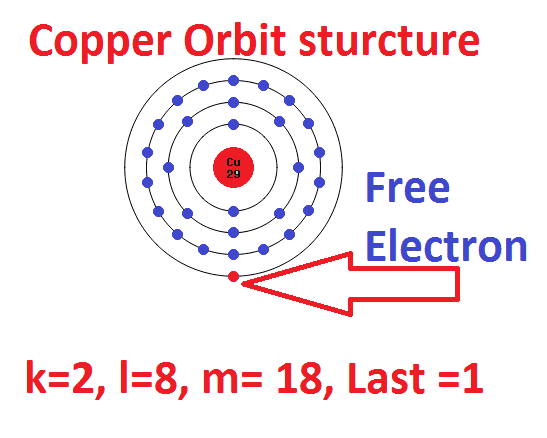
The subshell in which the last electron enters represent the block in which the element belongs. → What is the relation between the subshell to which the last electron was added and the block to which the element belongs? → What about the subshell to which the last electron of nitrogen was added 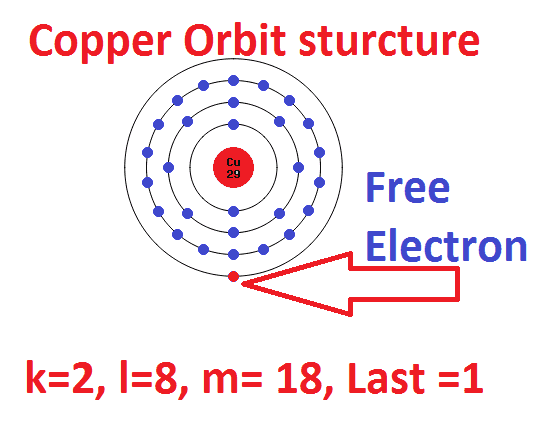
→ Which is the subshell of lithium to which the last electron was added? → How can the subshell electronic configuration be written in a short form? → What is the total number of electrons in the atom? →Which is the subshell to which the last electron was added? → How many shells are present in this atom? If the subshell wise electronic configuration of an atom is 1s 2 2s 2 2p 6 3s 2, find answers to the following: → On the basis of this, identify the correct electronic configuration of 29Cu from those given below:ġs 2 2s 2 2p 6 3s 2 3p 6 3d 9 4s 2 – Falseġs 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 1 – True → Write the subshell electronic configuration of 24CrĢ4Cr – 1s 2 2s 2 2p 6 3s 2 3p 6 3d 5 4s 1 → Using the symbol of neon, write the subshell electronic configuration of sodium? → Subshell electronic configuration of sodium? → Write its subshell electronic configuration. →Which is the noble gas preceding sodium (11Na)? 
→ Write the electronic configu ration of 22Ti, 23V, the two elements after Sc.Ģ2Ti - 1s 2 2s 2 2p 6 3s 2 3p 6 3d 2 4s 2 →The electronic configuration of scandium (2lSc) is → Write the subshell wise electronic configu-ration of potassium. → Write down the subshells in the increasing order of their energies. →Among the 3d & 4s subshells which has higher energy? Which one has lower energy?Īmong the 3s & 3p subshells which has higher energy? → Compare the energies of Is and 2s subshells. → How was the shell wise electronic configuration of potassium written? → Write the electronic configuration of Carbon → Write the electronic configuration of Boron → Complete the electronic configuration of beryllium? → Write the electronic configuration of Lithium (3Li) → Complete the subshell electronic configuration? → How many electrons are present in helium ( 2He)? → What may be the maximum number of electrons to be filled in the ‘p’ subshell? 
→ What is the maximum number of electronics that can be accommodated in the ‘s’? → Which subshell is common to all shells? What will be the number of subshells in the ‘M’ shell and ‘N’ → The ‘K’ shell, which is the first shell, has 1 subshell. → What is the maximum number of electrons that can be accommodated in the M Shell? → How many electrons are present in the M shell, the outermost shell of argon? → Write the electronic configuration of sodium and argon and complete the Table. → Is the group 1 element a metal or a nonmetal? → Atomic number of sodium is 11 Electronic configuration – 2,8,1 → What is the basis of classification of elements in the periodic table? Kerala State Syllabus 10th Standard Chemistry Solutions Chapter 1 Periodic Table and Electronic Configuration Periodic Table and Electronic Configuration Text Book Questions and Answers You can Download Periodic Table and Electronic Configuration Questions and Answers, Summary, Activity, Notes, Kerala Syllabus 10th Standard Chemsitry Solutions Chapter 1 help you to revise complete Syllabus and score more marks in your examinations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
